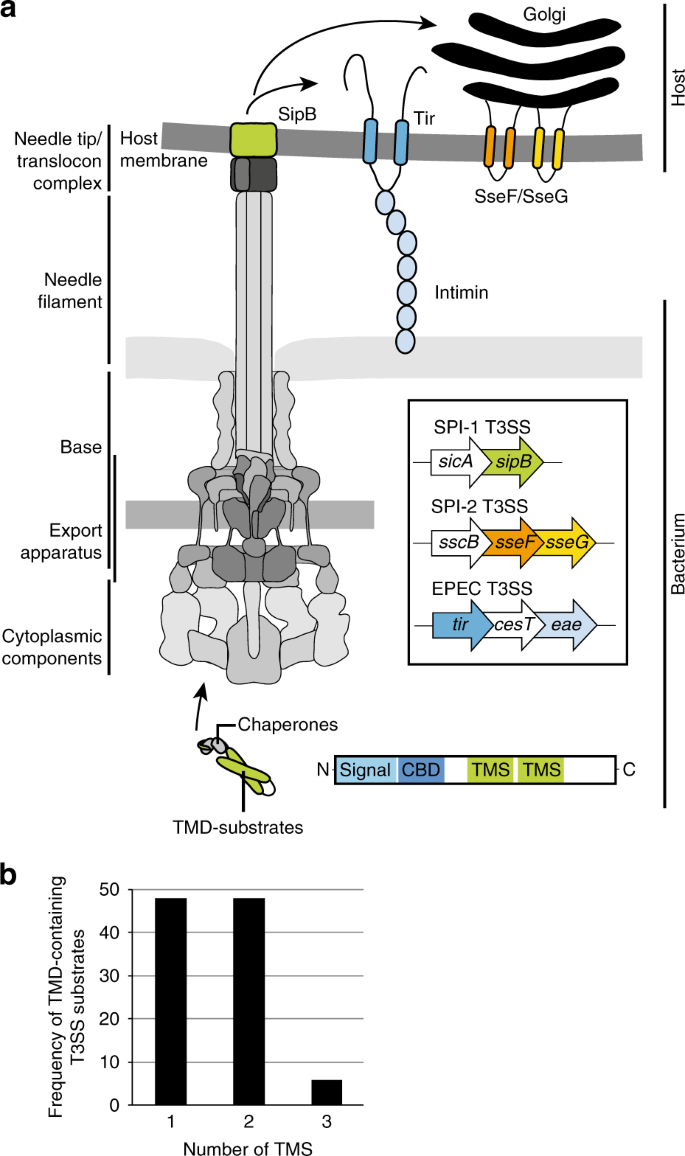
MemBrain: An Easy-to-Use Online Webserver for Transmembrane Protein Structure Prediction | SpringerLink
Membrane protein contact and structure prediction using co-evolution in conjunction with machine learning | PLOS ONE
MemBrain: An Easy-to-Use Online Webserver for Transmembrane Protein Structure Prediction | SpringerLink
MemBrain: An Easy-to-Use Online Webserver for Transmembrane Protein Structure Prediction | SpringerLink

Multiple C2 domains and transmembrane region proteins (MCTPs) tether membranes at plasmodesmata | EMBO reports

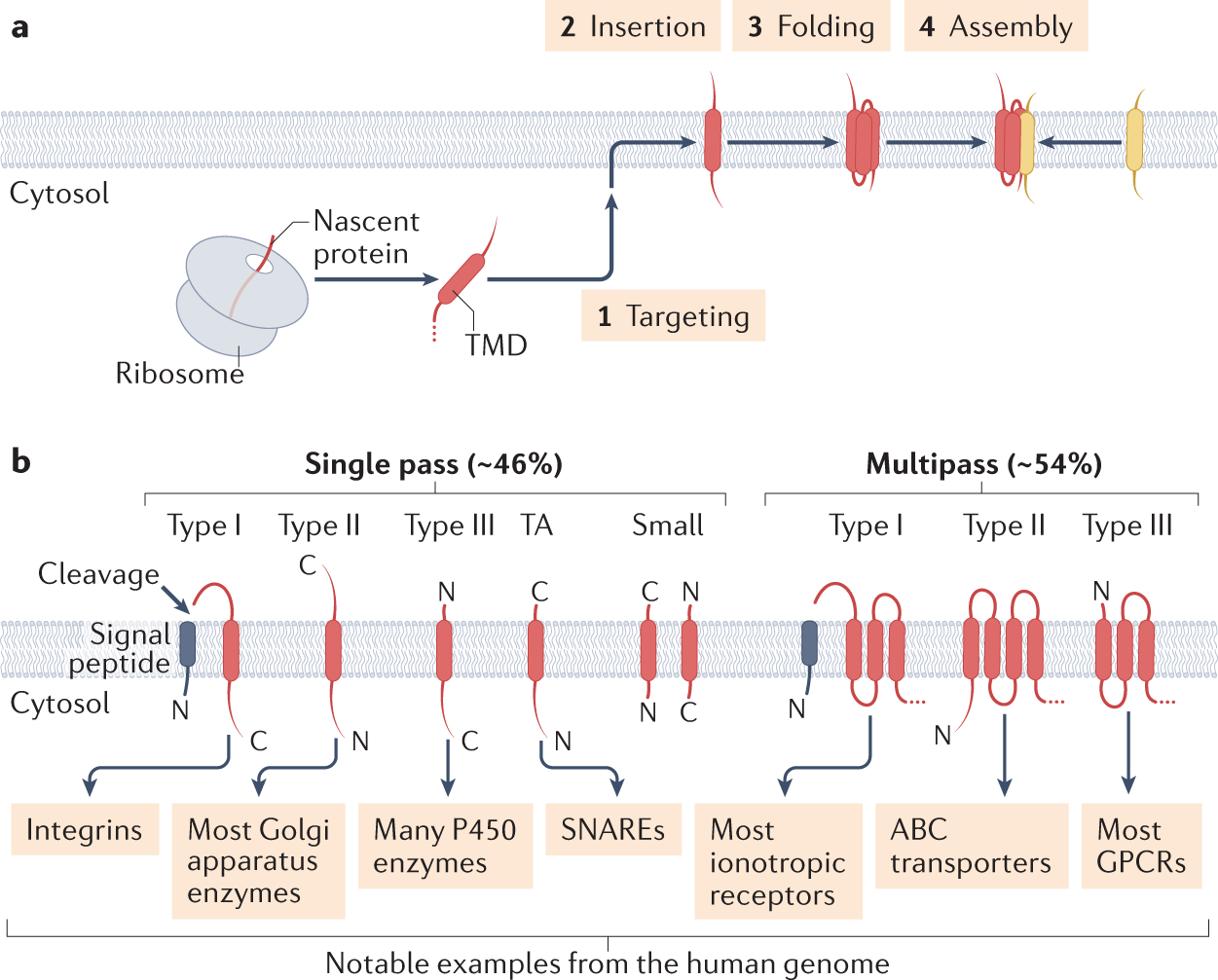
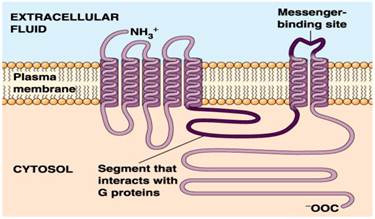
Interplay between hydrophobicity and the positive-inside rule in determining membrane-protein topology | PNAS

MutTMPredictor: Robust and accurate cascade XGBoost classifier for prediction of mutations in transmembrane proteins - ScienceDirect

Frontiers | A Gene-Based Positive Selection Detection Approach to Identify Vaccine Candidates Using Toxoplasma gondii as a Test Case Protozoan Pathogen
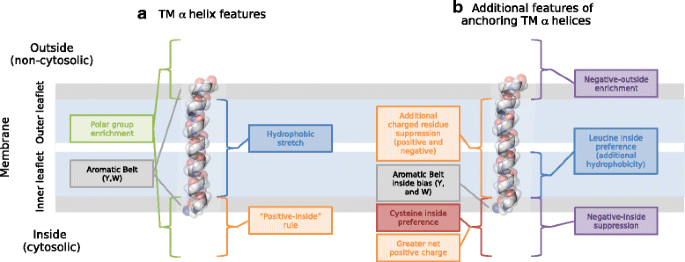
Charged residues next to transmembrane regions revisited: “Positive-inside rule” is complemented by the “negative inside depletion/outside enrichment rule” | BMC Biology | Full Text

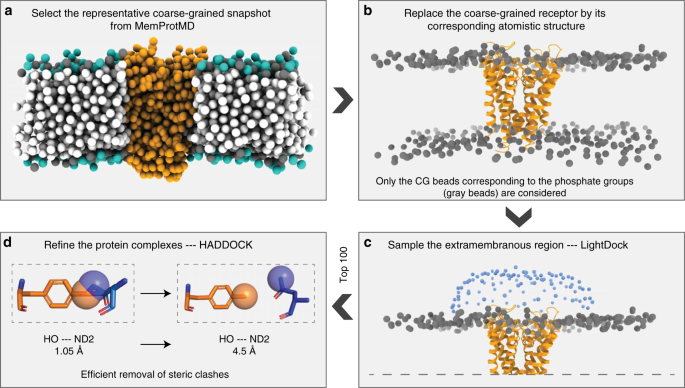
Getting to know each other: PPIMem, a novel approach for predicting transmembrane protein-protein complexes - ScienceDirect
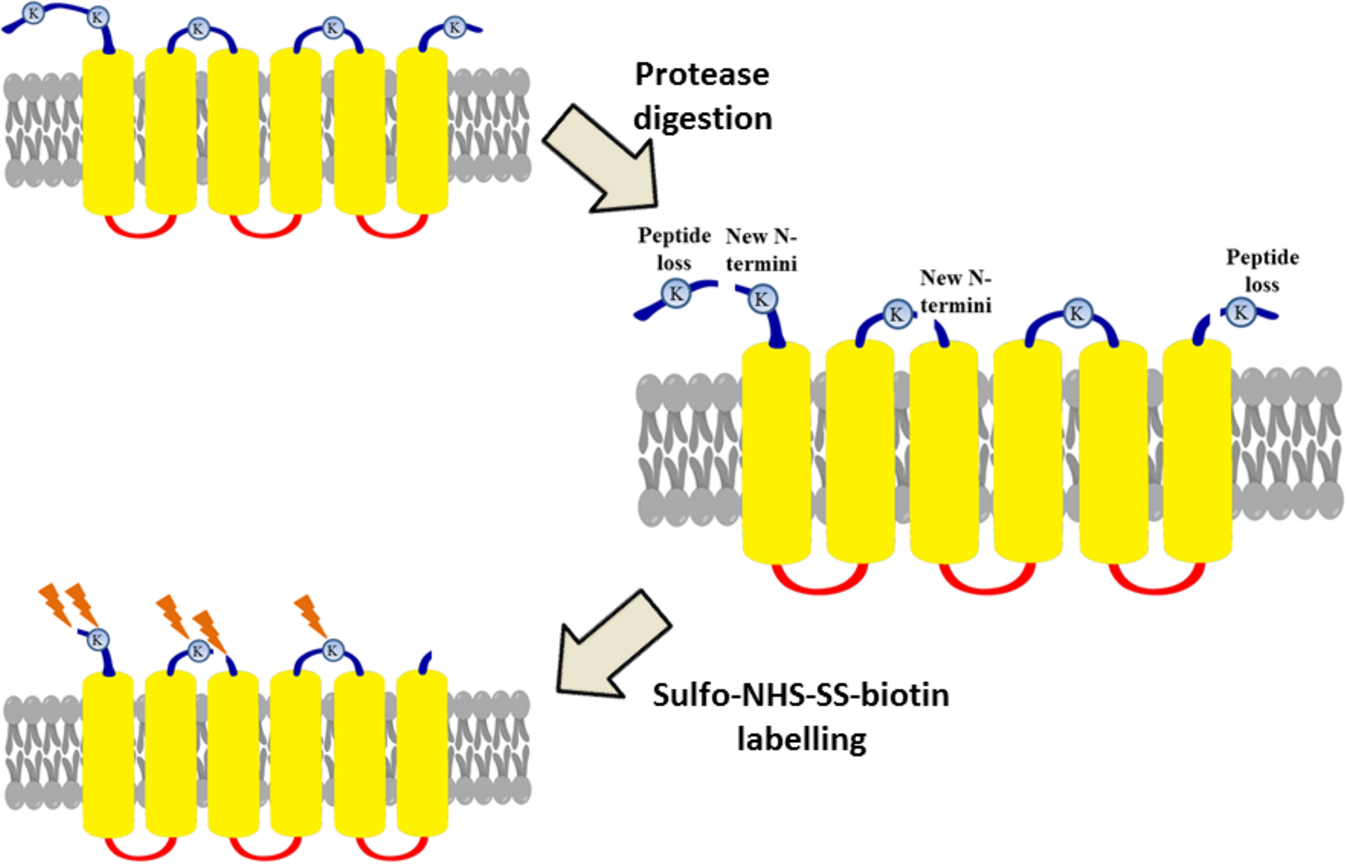
Partial proteolysis improves the identification of the extracellular segments of transmembrane proteins by surface biotinylation | Scientific Reports

Methods for Systematic Identification of Membrane Proteins for Specific Capture of Cancer-Derived Extracellular Vesicles - ScienceDirect
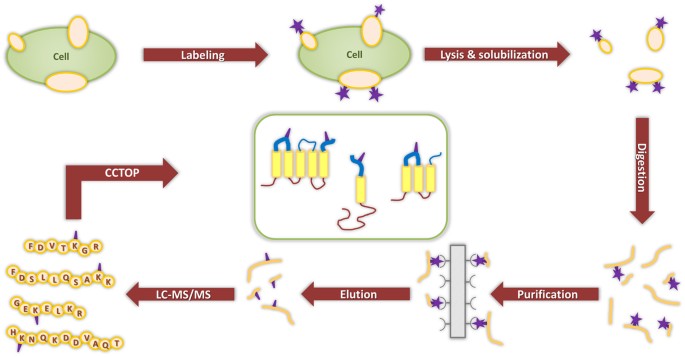
Identification of Extracellular Segments by Mass Spectrometry Improves Topology Prediction of Transmembrane Proteins | Scientific Reports